Find the missing value for the radioactive isotope – Unveiling the secrets of radioactive isotopes, this guide delves into the intricacies of their decay, half-life, and missing value calculations, unraveling their applications in diverse fields while emphasizing safety and regulatory considerations.
Understanding Radioactive Isotopes
Radioactive isotopes are atoms with an unstable nucleus, containing an excess of energy. This excess energy is released through the emission of radiation, leading to the transformation of the isotope into a more stable form. Radioactive isotopes are characterized by their atomic number, mass number, and half-life.
Radioactive isotopes are found naturally in the environment, such as uranium-238 and potassium-40. They can also be artificially produced in nuclear reactors or particle accelerators.
Properties and Characteristics of Radioactive Isotopes
- Unstable nucleus with excess energy
- Release radiation to achieve stability
- Characterized by atomic number, mass number, and half-life
- Can be naturally occurring or artificially produced
Examples and Applications of Radioactive Isotopes
- Uranium-238:Used in nuclear power plants and nuclear weapons
- Potassium-40:Used in geological dating and medical imaging
- Technetium-99m:Used in medical imaging for diagnosing various diseases
- Cobalt-60:Used in cancer therapy and industrial sterilization
Decay and Half-Life: Find The Missing Value For The Radioactive Isotope
Process of Radioactive Decay
Radioactive decay is the process by which an unstable radioactive isotope releases excess energy and transforms into a more stable form. This process involves the emission of radiation, such as alpha particles, beta particles, or gamma rays.
Concept of Half-Life, Find the missing value for the radioactive isotope
Half-life is the time it takes for half of the radioactive atoms in a sample to decay. It is a characteristic property of each radioactive isotope and is used to determine the rate of decay.
Formulas and Calculations Related to Decay and Half-Life
The decay constant (λ) of a radioactive isotope is related to its half-life (t 1/2) by the following formula:
λ = ln(2) / t1/2
The number of radioactive atoms remaining (N t) after a time t is given by:
Nt= N 0
e-λt
where N 0is the initial number of radioactive atoms.
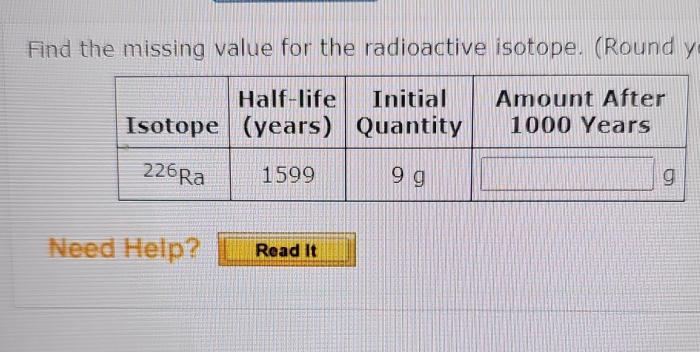
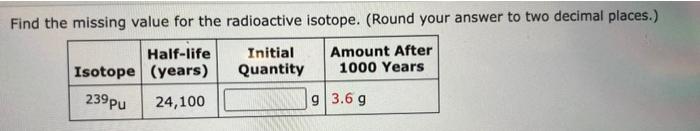
Missing Value Calculations
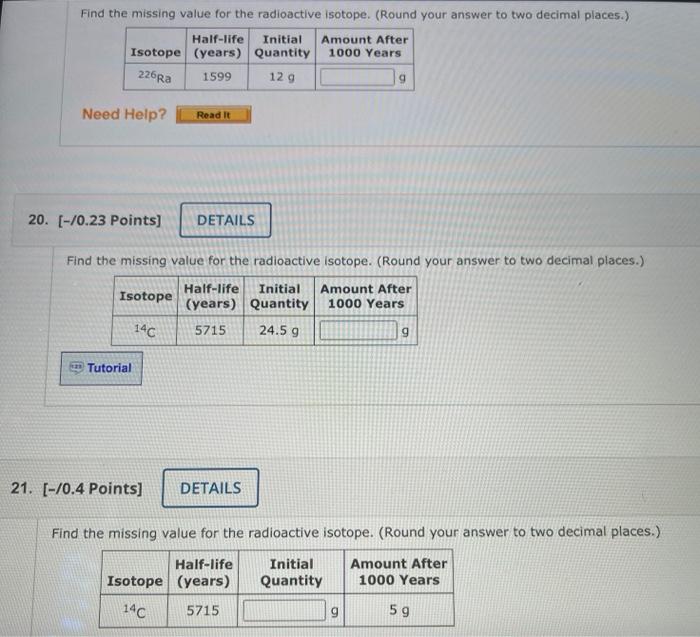
Methods for Calculating Missing Value
The missing value for a radioactive isotope can be calculated using various methods, including:
- Activity measurement:Measuring the radioactivity of a sample to determine the number of radioactive atoms present
- Half-life measurement:Determining the half-life of the isotope and using it to calculate the decay rate
- Isotopic ratio measurement:Comparing the abundance of different isotopes of the same element to determine the relative abundance of the radioactive isotope
Step-by-Step Guide for Solving Missing Value Problems
- Identify the available information, such as the half-life, activity, or isotopic ratio.
- Choose the appropriate calculation method based on the available information.
- Apply the relevant formulas and calculations to determine the missing value.
- Verify the results and ensure they are consistent with the given information.
HTML Table Illustrating the Calculation Process
| Method | Formula | Steps |
|---|---|---|
| Activity measurement | A = λN |
|
| Half-life measurement | t1/2 = ln(2) / λ |
|
| Isotopic ratio measurement | R = N1 / N2 |
|
Applications in Various Fields
Applications in Medicine
- Medical imaging:Using radioactive isotopes to visualize and diagnose various diseases, such as cancer and heart conditions
- Cancer therapy:Utilizing radioactive isotopes to target and destroy cancer cells
- Radiotherapy:Employing radioactive isotopes to treat certain types of cancer
Applications in Environmental Studies
- Radioactive dating:Determining the age of geological materials and fossils using radioactive isotopes
- Environmental monitoring:Tracking the movement and fate of pollutants in the environment
- Water resource management:Studying groundwater flow and contamination using radioactive isotopes
Applications in Industrial and Research
- Industrial sterilization:Using radioactive isotopes to sterilize medical devices and food products
- Material testing:Employing radioactive isotopes to analyze the structure and properties of materials
- Research:Utilizing radioactive isotopes to study various scientific phenomena, such as nuclear physics and biochemistry
Safety and Precautions
Safety Measures for Handling Radioactive Isotopes
- Shielding:Using lead or concrete barriers to protect against radiation exposure
- Distance:Maintaining a safe distance from radioactive sources
- Time:Minimizing exposure time to radiation
- Personal protective equipment:Wearing appropriate protective clothing, such as gloves, lab coats, and respirators
Regulations and Guidelines for Working with Radioactive Materials
- Licensing:Obtaining a license from regulatory authorities to handle radioactive materials
- Training:Receiving proper training in radiation safety protocols
- Monitoring:Regularly monitoring radiation levels and personnel exposure
- Emergency preparedness:Establishing procedures for handling accidents and spills
Tips for Safe Storage and Disposal of Radioactive Isotopes
- Secure storage:Keeping radioactive materials in locked, well-ventilated areas
- Proper disposal:Following established guidelines for disposing of radioactive waste
- Waste segregation:Separating different types of radioactive waste for proper management
- Record keeping:Maintaining accurate records of radioactive materials inventory and disposal
Answers to Common Questions
What is the significance of half-life in radioactive decay?
Half-life represents the time it takes for half of the radioactive atoms in a sample to decay, providing a crucial parameter for predicting the behavior and longevity of radioactive materials.
How are missing values calculated for radioactive isotopes?
Missing values can be determined using various methods, including the decay equation, which involves calculating the initial or final activity based on known values and the elapsed time.
What safety precautions should be taken when handling radioactive isotopes?
Stringent safety measures are paramount, including proper shielding, protective clothing, designated storage areas, and adherence to regulatory guidelines to minimize exposure and potential hazards.